A.H. Jay Burr, Associate Professor
SENSORY PHYSIOLOGY

A.B. Hamilton College
PhD. Rockefeller University
Room B8226,
(778) 782-4803/4284
burr@sfu.ca
SFU
Current Research Program
Many lower invertebrates negotiate their world with simple sensory responses such as avoidance reflexes and sensory orientation (taxis). However, the sensory structures and neural mechanisms underlying this behavior is largely unstudied. My lab is interested in simple eyes and light-controlled behavior in nematodes. These transparent worms have simple sensory organs and a very small but well-developed nervous system. Four different eye structures have evolved.
Mermis nigrescens, a parasite with an unusually large-sized free-living stage, is probably the best animal for studying mechanisms of phototaxis. A cyllindrical region of dense pigment shadows a photoreceptor when the head is not oriented towards a light source (see Fig. A).
Microspectrophotometry revealed that the pigment is oxyhemoglobin, although the absorbance spectrum is slightly unusual (Fig. B). This unique eye structure together with a scanning motion of the head provides a simple mechanism for orientation towards the brightest region of the environment. The neural signalling involved is still a mystery and we would like to investigate this using electrophysiological methods.
Mermis eye hemoglobin not only has a high oxygen affinity, it also has a unique shadowing role and is the only known naturally-crystalline hemoglobin (see dichroic spectrum, Fig. B). With other labs we are seeking its amino acid sequence for structural comparisons with other hemoglobins. The hemoglobin eye pigment appears late in the development of the female. We would like to determine if this is controlled by Pax-6, a proposed įmaster control geneš for eye development in animals, and a collaborator is now sequencing the Mermis homologue.
Many nematodes lack eyes altogether, however, this doesnšt rule out light sensitivity. We have discovered an avoidance response to light in Caenorhabditis elegans, a nematode being intensively studied as a model organism by laboratories around the world. Much is already known about its avoidance reflex to other stimuli and the neurons involved. However the photoreceptive neuron has yet to be identified. Using a computer-video tracking system developed in our laboratory, we are now narrowing the possibilities by testing existing mutants that have known defects in different subsets of the sensory neurons.
Selected Publications
Gans, C. and A.H.J. Burr. 1994. The unique locomotory mechanism of Mermis nigrescens, a large nematode which crawls over soil and climbs through vegetation. J. Morphol. 221: 1-16.
Burr, A.H.J., C.P.F. Babinszki. 1990. Scanning motion, ocellar morphology and orientation mechanisms in the phototaxis of the nematode M. nigrescens. J. Compar. Physiol. A. 167: 257-268.
Burr, A.H. 1985. The photomovement of Caenorhabditis elegans. A nematode which lacks ocelli. Proof that the response is to light not radiant heating. Photochem. Photobiol. 41: 577-582.
Burr, A.H. and F. Harosi. 1985. Naturally crystalline hemoglobin of the nematode Mermis nigrescens. Biophysical J. 47: 527-536.
Burr, A.H. 1984. Evolution of eyes in the lower phyla. In: M. A. Ali. (Ed.) Photoreception and Vision in Invertebrates. Plenum Press, New York. pp. 131-178.
Burr, A.H. 1984. Photomovement behavior in simple invertebrates. In: M. A. Ali. (Ed.) Photoreception and Vision in Invertebrates. Plenum Press, New York. 179-215.
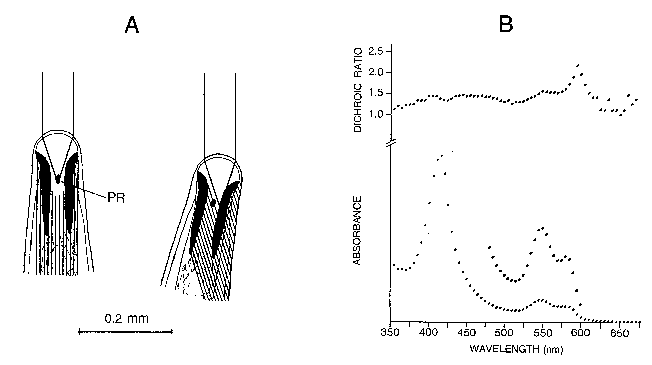 A) Eye of the nematode Mermis nigrescens, illustrating the shadowing of a phtoreceptor (PR) during the scanning motion.
A) Eye of the nematode Mermis nigrescens, illustrating the shadowing of a phtoreceptor (PR) during the scanning motion.
B) Spectral evidence that the shadowing pigment is a crystalline oxyhemoglobin.
This page last updated October 1996.

