research projects
To understand various aspects of the eukaryotic cytoskeleton and cilia, we carry out our research using a multidisciplinary approach that spans the fields of molecular biology, cell biology, biochemistry, genomics and genetics. We use a combination of in vitro experiments and in vivo studies with several model organisms, including C. elegans, mammalian cells and yeast.
Various research projects being pursued in our lab are summarised below.
cilia
Our lab has a general interest in understanding the makeup and various functions of cilia. Cilia are small appendages that protrude from the surface of many unicellular and essentially all multicellular organisms. 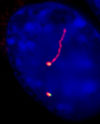 In humans, for example, cilia are found on most cell types. There are two classes of cilia; one is non-motile (typically called 'primary') and the other is motile and is interchangeably called flagella. Primary cilia (shown) are known to be generally involved in sensing the external environment and relaying messages to the associated cell. Sensory cilia perform a wide array of functions, including perceiving light (i.e., rods and cones are modified cilia), sensing chemicals (i.e., smell), and sensing fluid flow (e.g., in kidneys). Primary cilia also play an important role during development, as cilia at the embryonic node are involved in establishing the left-right symmetry of animals. Motile cilia play critical roles in several motility processes, such as sperm locomotion, and clearance of fluids and debris from the respiratory tract.
In humans, for example, cilia are found on most cell types. There are two classes of cilia; one is non-motile (typically called 'primary') and the other is motile and is interchangeably called flagella. Primary cilia (shown) are known to be generally involved in sensing the external environment and relaying messages to the associated cell. Sensory cilia perform a wide array of functions, including perceiving light (i.e., rods and cones are modified cilia), sensing chemicals (i.e., smell), and sensing fluid flow (e.g., in kidneys). Primary cilia also play an important role during development, as cilia at the embryonic node are involved in establishing the left-right symmetry of animals. Motile cilia play critical roles in several motility processes, such as sperm locomotion, and clearance of fluids and debris from the respiratory tract.
ciliary functional genomics
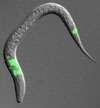 We use a number of bioinformatic, genomic and genetic strategies to uncover the components that make up cilia, mainly using C. elegans as the organism of choice. The expression of candidate ciliary genes in cilia-containing cells is verified (pictured; ciliated cells are near the nose and tail of the animal). In addition, the localisation and possible dynamic behaviour of proteins (see below) the ciliary genes encode is assessed by creating transgenic animals expressing the protein fused to GFP. The functions of novel genes are then probed by analysing cilia functions in mutant animals.
We use a number of bioinformatic, genomic and genetic strategies to uncover the components that make up cilia, mainly using C. elegans as the organism of choice. The expression of candidate ciliary genes in cilia-containing cells is verified (pictured; ciliated cells are near the nose and tail of the animal). In addition, the localisation and possible dynamic behaviour of proteins (see below) the ciliary genes encode is assessed by creating transgenic animals expressing the protein fused to GFP. The functions of novel genes are then probed by analysing cilia functions in mutant animals.
intraflagellar transport
 The biogenesis (formation) and maintenance of cilia requires a process termed intraflagellar transport, or IFT for short. IFT, which occurs bi-directionally along the microtubules that make up cilia, makes use of kinesin and dynein molecular motors to transport cargo needed for the function of cilia. Ciliary components accumulate at the base of cilia in a subcellular structure termed basal body (a modified centriole), and are then transported into the cilium. We use bioinformatics, functional genomics, cell biology and genetic experiments to uncover the various components making up the IFT machinery, and study the IFT process by carrying out time-lapse microscopy observations of GFP-tagged proteins in living C. elegans.
The biogenesis (formation) and maintenance of cilia requires a process termed intraflagellar transport, or IFT for short. IFT, which occurs bi-directionally along the microtubules that make up cilia, makes use of kinesin and dynein molecular motors to transport cargo needed for the function of cilia. Ciliary components accumulate at the base of cilia in a subcellular structure termed basal body (a modified centriole), and are then transported into the cilium. We use bioinformatics, functional genomics, cell biology and genetic experiments to uncover the various components making up the IFT machinery, and study the IFT process by carrying out time-lapse microscopy observations of GFP-tagged proteins in living C. elegans.
Bardet-Biedl syndrome, Meckel syndrome and other ciliopathies
Our lab has played an instrumental role in establishing Bardet-Biedl syndrome (BBS) as a disorder caused by the improper function of cilia (ciliopathy). BBS is of particular interest because it is characterised by a large number of different ailments, including obesity, blindness, kidney and heart anomalies, and neurosensory impairment (e.g., smell and hearing is attenuated in patients). Furthermore, there are now eight different genes known to be linked to BBS, and it is estimated that there may be at least as many that have not been identified. We have a general interest in ciliopathies. For example, cilia dysfunction can result in polycystic kidney disease, and various other genetic diseases likely result from improper cilia function (e.g., Alstrom syndrome). To understand some of the cellular problems associated with BBS, for example, we study the C. elegans homologues of the proteins linked to the disorder, and we also study these proteins using mammalian tissue culture cells. Using similar approaches, we are now dissecting the molecular functions of proteins implicated in Meckel syndrome.
Molecular chaperones
Our lab has had a long-standing interest in understanding the roles that molecular chaperones play in assisting the biogenesis of actin and tubulin, two cytoskeletal proteins that perform a multitude of essential functions in eukaryotes.  Two chaperones of particular interest include the chaperonin known as CCT or TRiC, and its co-chaperone, prefoldin. CCT interacts with and assists the folding of a significant proportion of nascent polypeptides. It is composed of eight related proteins that form two stacked rings with a central cavity wherein ATP-dependent folding takes place. Prefoldin is a hexameric protein complex consisting of 6 different polypeptides in eukaryotes (shown). It interacts with and stabilises unstable, newly-made forms of actin and tubulin (and likely other proteins), and transfers them to CCT for productive folding. We have recently uncovered two other cofactors of CCT, the phosducin-like proteins 2 and 3, which together with prefoldin are required for proper actin and tubulin biogenesis.
Two chaperones of particular interest include the chaperonin known as CCT or TRiC, and its co-chaperone, prefoldin. CCT interacts with and assists the folding of a significant proportion of nascent polypeptides. It is composed of eight related proteins that form two stacked rings with a central cavity wherein ATP-dependent folding takes place. Prefoldin is a hexameric protein complex consisting of 6 different polypeptides in eukaryotes (shown). It interacts with and stabilises unstable, newly-made forms of actin and tubulin (and likely other proteins), and transfers them to CCT for productive folding. We have recently uncovered two other cofactors of CCT, the phosducin-like proteins 2 and 3, which together with prefoldin are required for proper actin and tubulin biogenesis.
Contact us
Contact info, directions can be found here
[ page updated Nov. 11, 2015 ]